R & F® Listeria monocytogenes Chromogenic Plating Medium (Licensed from Biosynth AG)
Request a Quote

Documentation Links:
Certificate of Analysis
SDS
SDS Listeria monocytogenes agar
SDS Listeria monocytogenes agar supplement
SDS Listeria monocytogenes plates

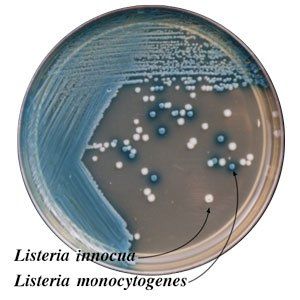
Colony Appearance

Mechanism
Industry Types

Advantages/Benefits

Publications

Ordering Information
M-0550
R & F® Listeria monocytogenes Chromogenic Detection System (250 g Bottle of Powder & two Supplement Boxes – Makes 220-240 Plates)
M-0500
R & F® Listeria monocytogenes Chromogenic Plating Medium (250 g Bottle)
M-0510
R & F® Listeria moncytogenes Supplement for Plating Medium (1 Box)
M-0520
R & F® Listeria moncytogenes Confirmatory Medium (100 g Bottle)
M-0560
R & F® Listeria monocytogenes Chromogenic Prepared Plate (Minimum Order of 20 Plates – Shelf life is 60 Days)
M-0570
R & F® Listeria monocytogenes Confirmatory Prepared Biplate (Minimum Order of 20 Plates – Shelf life is 60 Days)